PROJECT OVERVIEW
The "Polyvac Rubella Ag Rapid Test" was developed at the Center for Research and Production of Vaccines and Biologicals (POLYVAC).
Research Details
- TOPIC NAME Polyvac Rubella Ag Rapid Test
- Classification Product Under Development
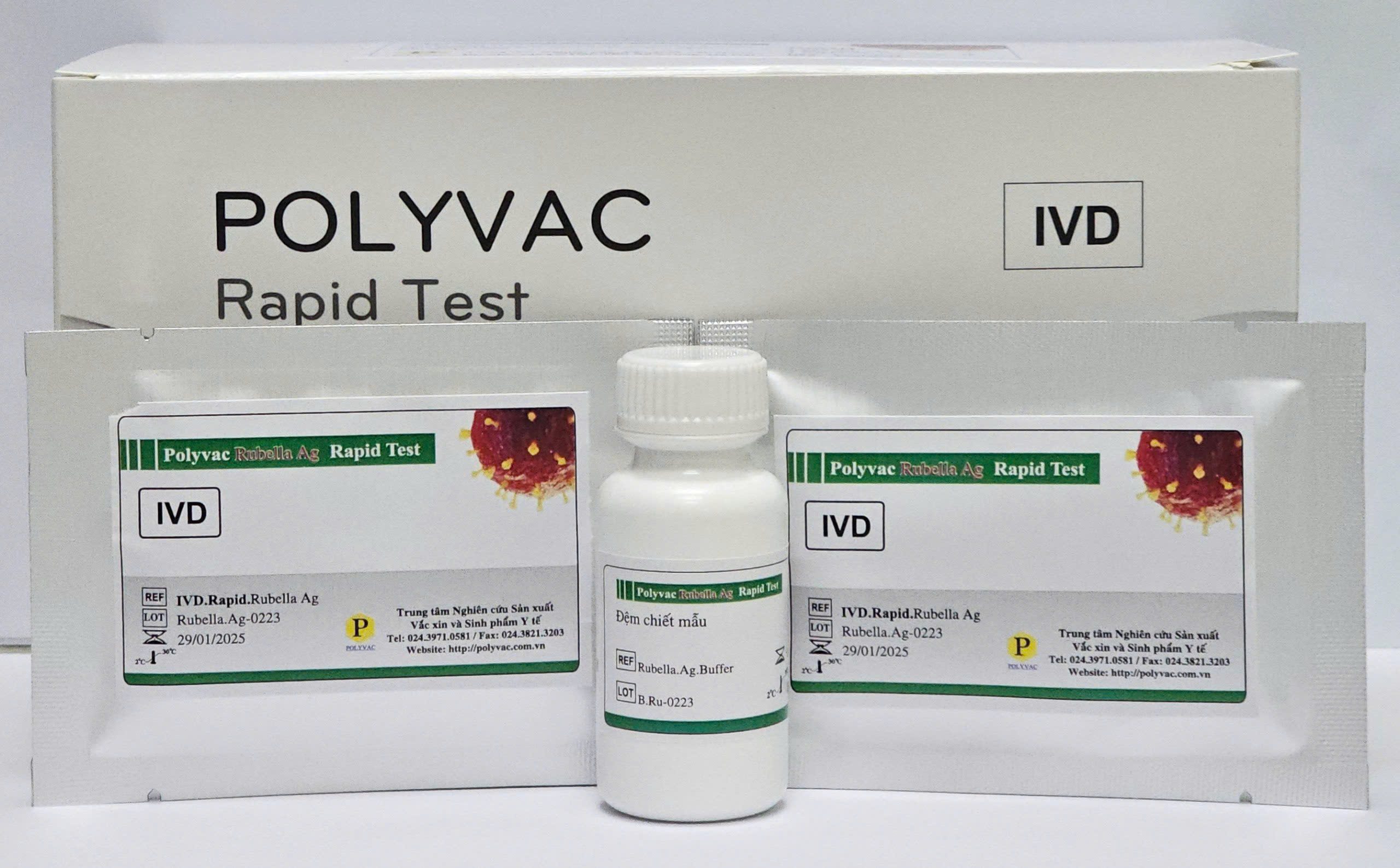
General Information
What is the Polyvac Rubella Ag Rapid Test?
The Rubella virus antigen qualitative test kit is based on immunochromatographic principles for the detection of Rubella virus in nasopharyngeal swab specimens. This test supports the diagnosis of Rubella virus infection.
Materials Required But Not Provided
- Timer
- Pen
- Gloves
- Face mask
Test Principle
Anti-Rubella virus antibodies are conjugated with gold nanoparticles and adsorbed onto the conjugate pad. Anti-Rubella virus antibodies are immobilized at the test line (T) position on the test cassette. Protein A is immobilized at the control line (C) position. When a sample containing Rubella virus is applied to the test cassette, a Rubella virus-anti-Rubella antibody-gold nanoparticle complex forms. This complex migrates along the nitrocellulose membrane by capillary action to the test line (T), where it is captured by anti-Rubella antibodies, forming an anti-Rubella antibody-Rubella virus-anti-Rubella antibody-gold nanoparticle complex that produces a signal line, indicating a positive result. The remaining conjugate (anti-Rubella antibody-gold nanoparticles) not captured by anti-Rubella antibodies migrates to the control line and is captured by Protein A, producing a signal line confirming normal test operation. Therefore, when both signal lines appear at the test line and control line, the sample is positive for Rubella virus. When only one signal line appears at the control position, the sample is negative for Rubella virus.
Test Procedure
- Use a pipette to transfer 1 ml of extraction buffer solution from the buffer bottle (28 ml) into the extraction tube.
- Insert a sterile swab into the patient's nostril, reaching the posterior nasopharyngeal surface.
- Gently rotate the swab on the posterior nasopharyngeal surface.
- Carefully remove the swab from the nasal cavity.
- Insert the nasopharyngeal swab into the extraction buffer tube. Rotate the swab at least five times.
- When removing the swab, squeeze both sides of the tube to extract all liquid from the swab.
- Securely attach the filter cap to the tube.
- Dispense 3 drops of sample (80-120 µl) into the sample well of the test cassette.
- Observe the result reading area of the test cassette. Read results within 5-10 minutes after sample application.
Result Interpretation
1. NEGATIVE: Only one line appears at position C. The result is Negative.
2. POSITIVE: When signals appear at both T and C positions in the result window (two lines), the result is Positive for Rubella virus.
3. INVALID: When no signal appears at position C in the result window.
Positive results should be interpreted in conjunction with clinical presentation and other available data by the physician.
Storage Conditions and Shelf Life
- Storage conditions: 2-30°C
- Shelf life: 24 months